What is Cardiomo and how it was started?
Cardiomo is an innovative device, app, and software solution for real-time monitoring of heart health, all conveniently packed into the palm of your hand.
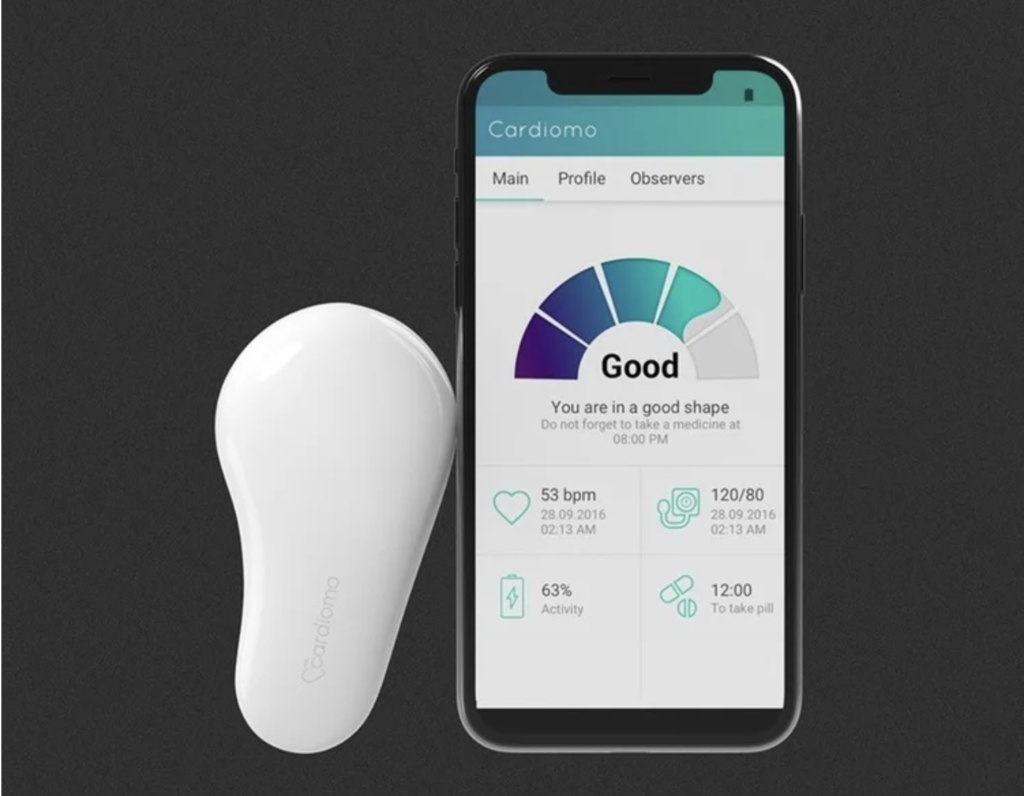
Here’s how it works: The Cardiomo gadget is attached to the left side of the chest using electrodes, where it reads essential heart indicators like electrocardiogram (ECG), heart rate, respiratory rate, and skin temperature. The data is then transmitted via Bluetooth to a mobile device running the Cardiomo application, available for both Android and iOS. From there, the information is seamlessly sent to cloud storage in real-time.
The Cardiomo device is supplied by a 280 mAh battery with an impressive lifetime of up to seven days on a single charge. It can be fully charged within three hours using the provided Type-C port, which features a silicone plug for protection. Designed to withstand water exposure, the device is waterproof and can even be worn while swimming, periodically synchronizing data with the mobile device by Bluetooth module every 30-60 minutes.
The startup behind Cardiomo is led by the husband-and-wife team of Roman Belkin and Ksenia Belkina. Ksenia, who holds a degree in psychology, oversees the management aspects, while Roman, a graduate of the Kharkiv Institute of Radio Electronics, focuses on technical development. This isn’t their first team project, as they have previously collaborated on various ventures, including a talking educational book for children and night driving glasses. Together, they have also successfully executed several large-scale social projects.

The idea for Cardiomo originated in 2014 when Roman Belkin wanted to assist his mother in remotely monitoring her health and heart function. The concept further solidified when Roman discovered a portable medical device used for Holter monitoring, which involves continuous recording of an electrocardiogram throughout the day. However, the traditional Holter monitor, with its Velcro electrodes and wired connections, proved inconvenient for long-term use. This prompted Roman to envision a device with holter-like functions that would be comfortable for extended wear, rechargeable, and integrated with artificial intelligence for online data analysis and transfer.
How Cardiomo works
Using biosensors, the Cardiomo device analyzes data to generate personalized health assessments and recommendations. It can provide insights on excessive or insufficient physical activity, stress levels, and relaxation periods. The device also offers alerts, including emergency notifications, such as detecting a fall or heart attack. Messages are delivered through the Cardiomo application on the user’s mobile device or their relatives’ devices. Roman assures that the messages are color-coded, resembling traffic lights: green for safe, yellow for lower, and red for critical levels of danger.
The device itself connects with the caregivers or relatives smartphone via Bluetooth module, giving an opportunity to monitor patient’s condition in real time. To connect device with your phone, you have to download the Cardiomo app. The app shows breathing rate, skin temperature, ECG, and the most important: heartbeat rate and blood pressure. If one of these indexes become critical, the device sends a push-notification to warn the caregiver, and even able to call an emergency itself. In addition, the app reminds to take medicines, so it is recommended to use for older people.
Cardiomo's path and future plans. Is Cardiomo a medical product?
During the development of their product and acquaintance with the market of medical gadgets, the developers came to the conclusion that their gadget is a medical product. After choosing such a development vector, Cardiomo sales had to be stopped – now startuppers need a medical certificate to implement their development. ISO 13485 certification, an international standard that regulates the quality management system for manufacturers of medical devices, machinery and equipment, Cardiomo already received in 2020. According to Belkin, the Cardiomo startup is preparing to receive Ukrainian certification next week, after which pilot sales in Ukraine begin – a preliminary contract has already been signed with a distributor for some of the devices from the second batch. As for European certification, it is obtained according to the new EU MDR standard. This is a new regulation on the certification of medical devices and equipment within the European Union, concerning organizations whose medical devices are exported to the EU countries.

Cardiomo has now passed the first phase of the European certification and is expected to pass the second phase in November. Cardiomo plans to obtain European CE Mark certification according to new standards by the end of 2021. For certification for the American market, by the end of 2021, it is planned to submit documents to the FDA for 510 (k) – this is the first stage of registration of medical devices in the United States for mandatory registration of manufacturers of medical devices, without which the import of medical devices into the United States is not allowed. Most of the documents submitted for European certification are also suitable for American certification, so the main package will be ready by the end of this year.
Ukrainians with their project have already visited Lithuania, China, Japan at the invitation of the host parties, where they presented Cardiomo at exhibitions and competitions. Their next step is Las Vegas.
Frequently Asked Questions (FAQ)
Cardiomo’s main task is to monitor heartbeat rate, blood pressure, breathing rate and even body temperature.
Through years, Cardiomo received a special international standart ISO 13485, officially making it a medical product.
Cardiomo can be used anywhere, even in the pool. It is completely waterproof. Small size of device gives you to use it freely, with the battery capacity of 280 mAh, Cardiomo can be used for a whole week without charging the battery. The battery itself charges quite quickly: 3 hours and your device is ready to work!
Infosoft is a team of IT and QA engineers. We provide companies with technical talents and product development experience to create world-class software. You can scale up and down your remote software developing team at any time without any financial risk.


